InnerPlant takes ‘meaningful step’ in regulatory journey
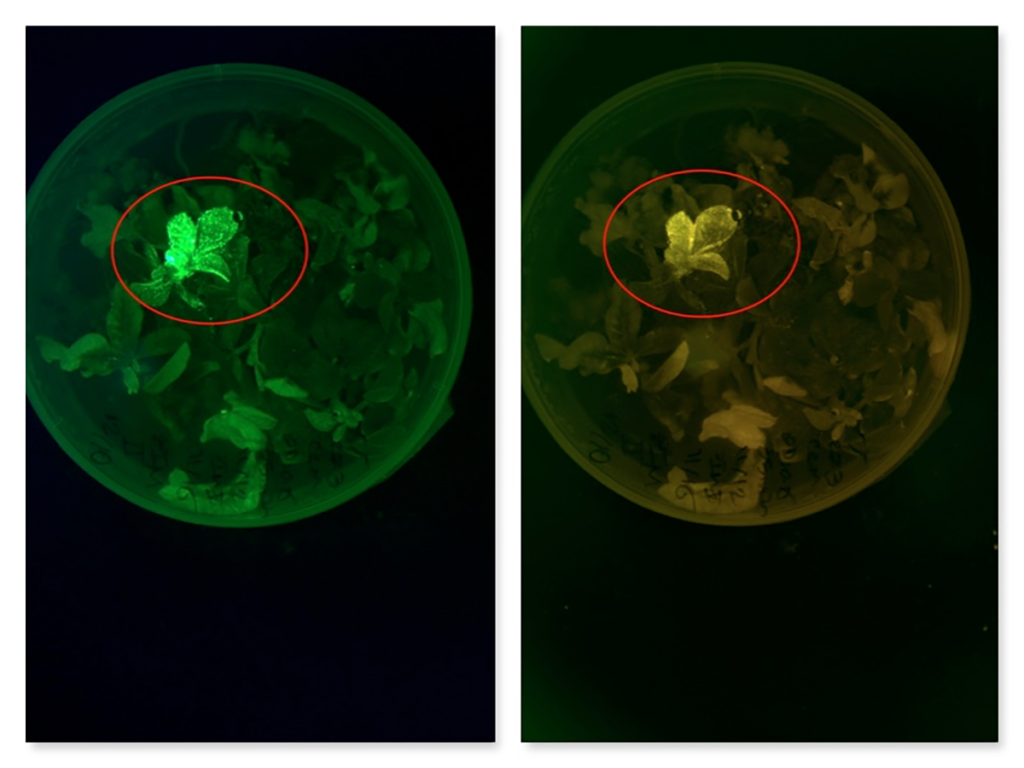
InnerPlant has successfully completed the FDA’s New Protein Consultation process for its InnerSoy product, which uses fluorescent proteins to signal plant distress. This milestone is crucial for advancing regulatory approval, enabling broader adoption of genetically engineered sensor plants that enhance agricultural efficiency and pest management.
Original excerpt: InnerPlant has completed the FDA’s New Protein Consultation (NPC) process for the fluorescent protein used in its InnerSoy product…
Source: AgFunderNews

Responses